Contents
- The COVID - 19 Vaccines
- The 3 COVID - 19 Vaccines
- Side Effects
- Priority Groups
- How They Work
- NHS and UK Government Plan
- Frequently Asked Questions
- Conclusion
- References
The COVID - 19 Vaccines
Right now on the UK market, there are 3 released, and approved by the Medicines and Healthcare products Regulatory Agency (MHRA), COVID - 19 vaccines as of Jan 2021. The first being the Pfizer-BioNTech vaccine, next the Oxford-AstraZeneca vaccine and the recently approved (8 January 2021 [1]) Moderna Vaccine. There are 2 more vaccines that are currently in development and being approved, the Novavax jab and another vaccine developed by Johnson & Johnson [2].
You can track the development of other COVID - 19 vaccines here.
A Vaccine, as defined by the World Health Organisation (WHO), is
"A simple, safe, and effective way of protecting people against harmful diseases, before they come into contact with them. It uses your body’s natural defenses to build resistance to specific infections and makes your immune system stronger.” [3]
Vaccines are a part of life, in the UK you can have vaccines to protect yourself from a number of diseases. We have a national and childhood specific immunisation programme via the NHS which is rolled out to minimise the infections and outbreaks caused by vaccine preventable diseases, it is a cornerstone of public health and very important to the safety of our population. The childhood immunisation programme runs from 0 - 18 and covers a whole plethora of diseases [4]:
- Flu
- Diphtheria
- Tetanus
- Pertussis (whooping cough)
- polio (IPV)
- Haemophilus influenzae type b
- Hepatitis B
- Measles
- Mumps
- Rubella
- Meningitis (groups A, C, W, B and Y)
- Pneumococcal infection (PCV)
- human papillomavirus (HPV)
The 3 COVID - 19 Vaccines[5]:
Table 1: Summarised information of the 3 Vaccines | Source: Sky News, The Telegraph| Company | Doses Ordered by the UK (per million) | Cost per Dose (£) | Efficacy (%) | Storage (°C) | Dosage Interval (days) |
|---|---|---|---|---|---|
| Pfizer-BioNTech | 40 | 15 | 95 | - 70 | 21* |
| Oxford-AstraZeneca | 100 | 3 | 70 | 2 - 8 | 28* |
| Moderna | 7 | 28 | 94 | - 20 | 28 |
* When the Pfizer vaccine was initially approved and released, UK medicine regulators said the second dose of the Pfizer vaccine should be administered 21 days after the first - but this changed to a longer interval of between 21 days and 12 weeks. For the Oxford vaccine, the second dose should be taken between 4 and 12 weeks after the first dose.
The results above are based on studies completed by the corresponding companies to find the efficacy of their vaccines. Pfizer used a sample of over 40,000 people aged 18 - 65+ [6]. 30,000 for Moderna [7] and 23,000 between Brazil and the UK [8] for the Oxford - AstraZeneca vaccine.
There have been recent studies showing a new strain of the COVID - 19 virus however, early research shows that Pfizer and Moderna protect against the new strain [9].
Side Effects
You can find all the side effects in the government Green Book here, which documents all the side effects currently known.
Common side effects from the vaccine are as follows [10]:
- Tenderness, pain, warmth, redness, itching, swelling or bruising at the injection site
- Generally feeling unwell
- Feeling tired (fatigue)
- Chills or feeling feverish
- Headache
- Feeling sick (nausea)
- Joint pain or muscle ache
Some of these side effects are similar to the COVID - 19 symptoms as they are due to your immune system’s response to the virus, the key difference between distinguishing whether your symptoms are due to COVID - 19 or the vaccine are that the vaccine side effects only last a few days.
If you have received the vaccine and think you have side effects that have not been recorded, then use the Yellow Card website or the MHRA Yellow card app on the Google Play Store or the Apple Store to record your side effects to be tested and recorded, ensuring safe and effective use for others.
Priority Groups
The current priority groups will be given the vaccine in the following order:
- 80 year-olds and over and frontline health and social care workers
- 75 year-olds and over
- 70 year-olds and over and clinically extremely vulnerable individuals
- 65 year-olds and over
- 16 to 64 year-olds with serious underlying health conditions
- 60 year-olds and over
- 55 year-olds and over
- 50 year-olds and over
These groups, as deemed by the UK government and The Joint Committee on Vaccination and Immunisation, are at the highest risk of becoming seriously ill from the COVID - 19 virus and therefore are the first to receive the vaccine.
How They Work
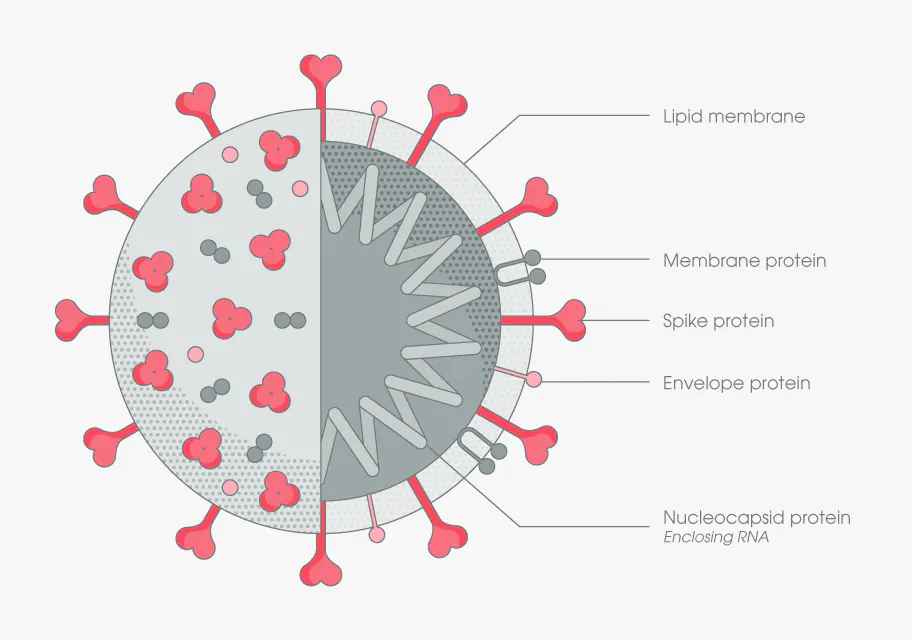
Pfizer-BioNTech and Moderna (BNT162b2 and mRNA-1273)
The Pfizer and Moderna COVID - 19 vaccines both work using the exact same mechanism. They both contain mRNA (Messenger Ribonucleic Acid), which is the genetic sequence required to build any protein in a living organism. These vaccines contains the mRNA of the spike protein found on the surface of the COVID - 19 virus (there is no whole or live virus inside the vaccine and therefore, it cannot cause disease). This mRNA is then wrapped in a lipid envelope (nanoparticle) which enables it to be transported safely into the cells of the body [11].
Once this vaccine is injected, the mRNA is engulfed by the host’s cells, which then translate the genetic information stored in the mRNA using the cell’s ribosomes to produce spike proteins and thus never even touching the nucleus or any part of the interior of the cell. These spike proteins are then expressed onto the surface of the cell, which in turn stimulates the immune system via T - cells to generate more T - cells and B - cells which produce antibodies and prepare the immune system to respond to any further exposure to the COVID - 19 virus by disabling the virus or binding the virus together through the process of agglutination and the generation of memory cells. Memory cells can remember the COVID - 19 virus and can therefore produce a more rapid immune response when a secondary infection is detected, this secondary infection is removed many times faster due to the memory cells.
The mRNA will then naturally degrade over the next few days.
Oxford-AstraZeneca COVID-19 vaccine (ChAdOx1 nCoV-19)
The AstraZeneca COVID - 19 vaccine uses a viral vector instead. This vector is a weakened chimpanzee adenovirus, and it acts as a carrier to deliver the COVID - 19 spike protein DNA. The adenovirus has been genetically modified to be non - replicating in human cells and therefore cannot cause disease. The genetic sequence of the spike protein, taken from the DNA of the COVID - 19 virus, is inserted into the adenovirus’s genetic code. When this vaccine is injected into the host, it enters the host’s cells which then use this DNA to produce the spike protein [12]. It then begins to work using the same mechanism as the Pfizer and Moderna vaccines, where the immune system is activated and starts to react by producing B - cells and T - cells which can disable the virus, produce antibodies and create memory cells for a future infection.
NHS and UK Government Plan
The UK government and NHS are deploying nationwide vaccinations to generate an acquired immunity population, which will protect the majority of people from the virus and help us get back to normality through herd immunity. If all members of the public have acquired immunity and create herd immunity, then it can exponentially decrease the number of people that can either become ill from the virus or even spread it by lowering the R value.
What is Acquired Immunity
Acquired immunity is the body’s 2nd line of defence. The immune system learns to recognise specific pathogens and challenge them if they meet them again later on.
What is Herd Immunity
When most of a population is immune to an infectious disease, this provides indirect protection, or herd immunity (also called herd protection), to those who are not immune to the disease [13].
Frequently Asked Questions
If there are vaccines with 90%+ efficacy, why use the Oxford Vaccine?
- The vaccine can be stored at 2-8 °C, which is standard fridge temperature, making it easier to store, transport and use
- It is the lowest costing vaccine of the 3, costing £3 per dosage
- It produces a good immune response from the most vulnerable groups, 65+
What is the difference between Pfizer and Moderna?
While they are both mRNA vaccines, the dosage interval for Moderna is 28 days while for Pfizer, it is 21. Each dose of Pfizer’s contains 0.3 ml of the vaccine while Moderna contains a larger dose of 1 ml.
How does mRNA vaccine differ from the Oxford Vaccine?
The Pfizer and Moderna vaccines use mRNA of the spike protein to produce an immune response while the Oxford vaccine uses a non - replicating chimpanzee adenovirus to transport the spike proteins DNA into the host to generate an immune response.
Do these vaccines change your DNA?
No, according to research, none of these vaccines alter your DNA or genetic information [14].
Conclusion
I am not a professionally trained expert on the vaccines, however, I have been training and volunteering with St Johns Ambulance in partnership with the NHS in administering the vaccine and, in my Biomedical Engineering degree, I have had the liberty to do some research on this topic.
I want to provide easily accessible information for others so they don’t worry about the vaccine as much, there are many conspiracy theories out there and every individual has a right to trust whatever they want, this blog post is just a way to summerise information about the vaccines and reduce worries about the vaccine and demonstrate how the NHS and the frontline workers out there doing their best to protect the health of the population.
And there have been benefits to this vaccine development apart from health, the mRNA technology was successfully tested and has accelerated the development of vaccines [15] which can prove to be very useful in future research. Also, as it only uses the spike protein, this vaccine is much safer compared to previous vaccines as there is no risk of actually getting the disease from the vaccine [16].
For more information on the virus and the vaccines, visit the the COVID - 19 Green Book here.
References
[1] Roberts, M. (2021). Moderna becomes third Covid vaccine approved in the UK. [online] BBC News. Available at: https://www.bbc.co.uk/news/health-55586410 [Accessed 23 Jan. 2021]
[2] Gallagher, J. (2021). Covid vaccine: Single-dose Johnson & Johnson jab is 66% effective. [online] BBC News. Available at: https://www.bbc.co.uk/news/health-55857530 [Accessed 23 Jan. 2021]
[3] Who.int. (2018). Vaccines and immunization: What is vaccination? [online] Available at: https://www.who.int/news-room/q-a-detail/vaccines-and-immunization-what-is-vaccination [Accessed 21 Jan. 2021]
[4] nidirect. (2017). Childhood immunisation programme | nidirect. [online] Available at: https://www.nidirect.gov.uk/articles/childhood-immunisation-programme [Accessed 21 Jan. 2021]
[5] Binding, L. (2020). COVID-19 vaccines: How do the Moderna, Pfizer and Oxford coronavirus jab candidates compare? [online] Sky News. Available here[Accessed 21 Jan. 2021]
[6] Pfizer.com. (2020). Pfizer and BioNTech Conclude Phase 3 Study of COVID-19 Vaccine Candidate, Meeting All Primary Efficacy Endpoints | Pfizer. [online] Available here [Accessed 22 Jan. 2021]
[7] COVID-19 Clinical Research Coalition. (2020). Moderna | COVID-19 Clinical Research Coalition. [online] Available at: http://covid19crc.org/covid-19-studies/moderna/ [Accessed 23 Jan. 2021]
[8] Fidler, B. (2020). AstraZeneca, Oxford vaccine prevents COVID-19, though study results raise questions. [online] BioPharma Dive. Available at: https://www.biopharmadive.com/news/astrazeneca-oxford-coronavirus-vaccine-efficacy-results/589542/ [Accessed 23 Jan. 2021]
[9] Roberts, M. (2021). Moderna vaccine appears to work against variants. [online] BBC News. Available at: https://www.bbc.co.uk/news/health-55797312 [Accessed 24 Jan. 2021]
[10] Vaccine safety and the management of adverse events following immunisation. (2012). [online] . Available here [Accessed 24 Jan. 2021]
[11] Sanjay Mishra (2020). How mRNA vaccines from Pfizer and Moderna work, why they’re a breakthrough and why they need to be kept so cold. [online] The Conversation. Available here [Accessed 24 Jan. 2021]
[12] Gallagher, J. (2020). Coronavirus: Oxford vaccine triggers immune response. [online] BBC News. Available at: https://www.bbc.co.uk/news/uk-53469839 [Accessed 25 Jan. 2021]
[13] Lindsay Smith Rogers and JH Bloomberg School of Public Health (2020). What is Herd Immunity and How Can We Achieve It With COVID-19? [online] Johns Hopkins Bloomberg School of Public Health. Available at: https://www.jhsph.edu/covid-19/articles/achieving-herd-immunity-with-covid19.html [Accessed 25 Jan. 2021]
[14] Forster, V. (2021). Covid-19 Vaccines Can’t Alter Your DNA, Here’s Why. Forbes. [online] 12 Jan. Available at: https://www.forbes.com/sites/victoriaforster/2021/01/11/covid-19-vaccines-cant-alter-your-dna-heres-why/ [Accessed 25 Jan. 2021]
[15] Solis-Moreira, J. (2020). How did we develop a COVID-19 vaccine so quickly? [online] Medicalnewstoday.com. Available at: https://www.medicalnewstoday.com/articles/how-did-we-develop-a-covid-19-vaccine-so-quickly#mRNA-technology [Accessed 26 Jan. 2021]